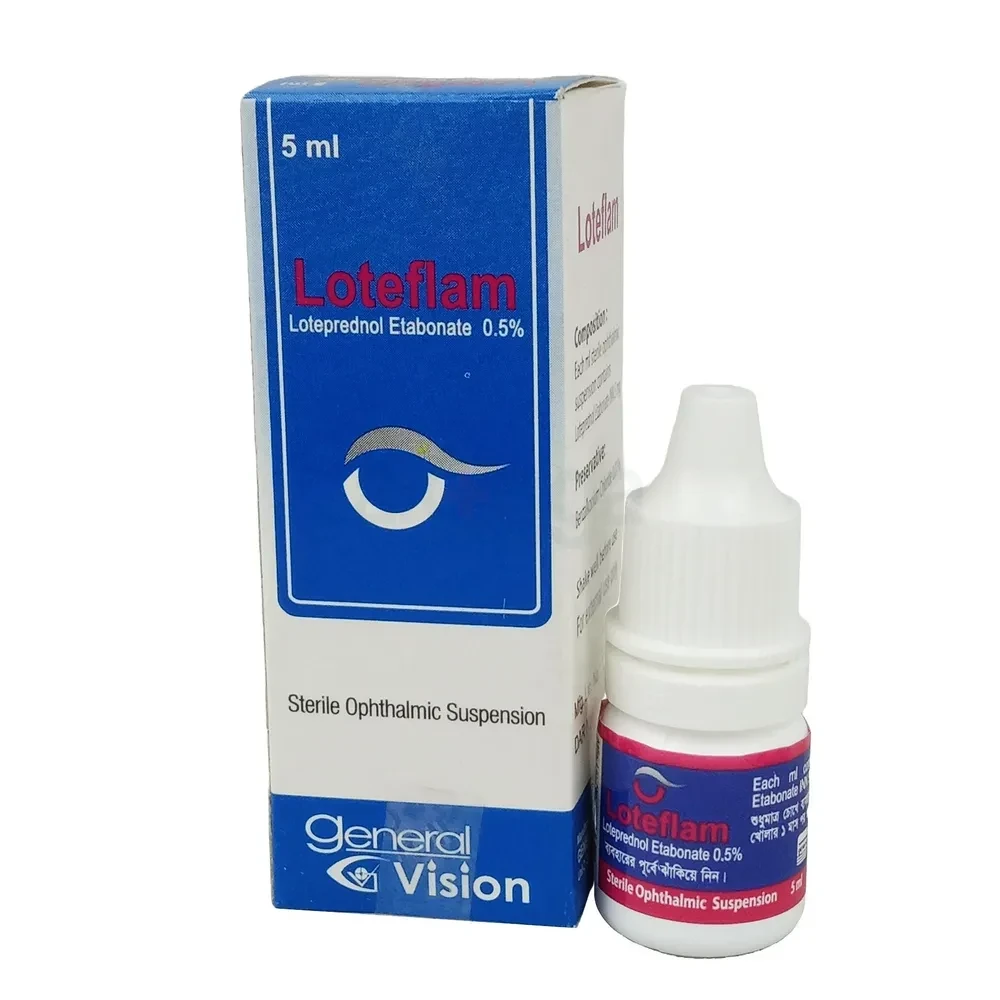

Loteflam Ophthalmic Suspension 5 ml drop, Loteprednol Etabonate 0.5%
Inhouse product
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00
Reviews & Ratings
Indications
0.5% ophthalmic
suspension: Loteflam suspension
is indicated in steroid responsive inflammatory conditions of the palpebral and
bulbar conjunctiva, cornea and anterior segment of eyeball such as allergic
conjunctivitis, acne rosacea, superficial punctate keratitis, herpes zoster keratitis,
iritis & cyclitis. It is also effective in post-operative inflammation.
1% ophthalmic
suspension: Loteflam suspension
is indicated in steroid responsive inflammatory conditions of the palpebral and
bulbar conjunctiva, cornea and anterior segment of eyeball such as allergic
conjunctivitis, acne rosacea, superficial punctate keratitis, herpes zoster
keratitis, iritis & cyclitis. It is also effective in post-operative
inflammation.
0.5% ophthalmic
ointment: Loteflam ointment is
indicated for the treatment of steroid responsive inflammatory conditions of
the palpebral and bulbar conjunctiva, cornea and anterior segment of the globe
such as allergic conjunctivitis, acne rosacea, superficial punctate keratitis,
herpes zoster keratitis, iritis, cyclitis, uveitis. Loteflam is also indicated
for the treatment of post-operative inflammation and pain following ocular
surgery.
0.5% Ophthalmic gel: Loteflam gel is indicated for the treatment
of post-operative inflammation and pain following ocular surgery. It is also
effective in steroid responsive inflammatory conditions of the palpebral and
bulbar conjunctiva, cornea and anterior segment of eyeball such as allergic
conjunctivitis, acne rosacea, superficial punctate keratitis, herpes zoster
keratitis, iritis & cyclitis.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Pharmacology
Corticosteroids
inhibit the inflammatory response to a variety of inciting agents and probably
delay or slow healing. They inhibit the edema, fibrin deposition, capillary
dilation, leukocyte migration, capillary proliferation, fibroblast
proliferation, deposition of collagen and scar formation associated with
inflammation. There is no generally accepted explanation for the mechanism of
action of ocular corticosteroids. However, corticosteroids are thought to act
by the induction of phospholipase A 2 inhibitory proteins, collectively called
lipocortins. It is postulated that these proteins control the biosynthesis of
potent mediators of inflammation such as prostaglandins and leukotrienes by
inhibiting the release of their common precursor arachidonic acid. Arachidonic
acid is released from membrane phospholipids by phospholipase A2.
Dosage & Administration
0.5% ophthalmic
suspension:
- For steroid responsive ocular
inflammation: Instill 1 drop of Loteprednol Ophthalmic Suspension into the
conjunctival sac of the eye(s) four times a day.
- For inflammation after surgery:
Instill 1 drop of Loteprednol Ophthalmic Suspension into the conjunctival
sac of the operated eye(s) four times a day beginning 24 hours after
surgery and continuing throughout the first two weeks after surgery.
1%
ophthalmic suspension:
- For steroid responsive ocular
inflammation: Instill 1 drop of Loteprednol DS Ophthalmic Suspension into
the conjunctival sac of the eye(s) 2 times a day.
- For inflammation after surgery:
Instill 1 drop of Loteprednol DS Ophthalmic Suspension into the
conjunctival sac of the operated eye(s) 2 times a day beginning 24 hours
after surgery and continuing throughout the first two weeks after surgery.
0.5%
ophthalmic ointment: Apply a small amount
(Approximately 1/2 inch ribbon) into conjunctival sac(s) four times daily
begining 24 hours after surgery and continuing throughout the first 2 weeks of
the postoperative period.
Ophthalmic gel: Instill one to two drops of Loteprednol Gel
Sterile Ophthalmic Gel into the conjunctival sac of the affected eye four times
daily beginning the day after surgery and continuing throughout the first 2
weeks of the postoperative period.
Use in children: Safety and effectiveness in pediatric
patients have not been established.
Geriatric Use: No overall differences in safety and
effectiveness have been observed between elderly and younger patients.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Interaction
Since Loteflam is not
detected in plasma following the topical administration, it is not expected to
affect the pharmacokinetics of systemically administered medicinal products.
Contraindications
Loteprednol, as with
other ophthalmic corticosteroids, is contraindicated in most viral diseases of
the cornea and conjunctiva including epithelial herpes simplex keratitis
(dendritic keratitis), vaccinia, and varicella, and also in mycobacterial
infection of the eye and fungal diseases of ocular structures. It is also
contraindicated in individuals with known or suspected hypersensitivity to any
of the ingredients of this preparation and to other corticosteroids.
Side Effects
Reactions associated
with ophthalmic steroids include elevated intraocular pressure, which may be
associated with optic nerve damage, visual acuity and field defects, posterior
subcapsular cataract formation, secondary ocular infection from pathogens including
herpes simplex and perforation of the globe where there is thinning of the
cornea or sclera. Ocular adverse reactions occurring in 5%-15% of patients
treated with Loteflam sterile ophthalmic suspension. (0.2%-0.5%) in clinical
studies included abnormal vision/blurring, burning on instillation, chemosis,
discharge, dry eyes, epiphora, foreign body sensation, itching, injection and
photophobia. Other ocular adverse reactions occurring in less than 5% of
patients include conjunctivitis, corneal abnormalities, eyelid erythema,
keratoconjunctivitis, ocular irritation/pain/discomfort, papillae. Non-ocular
adverse reactions occurred in less than 15% of patients. These include
headache, rhinitis and pharyngitis.
Pregnancy & Lactation
Pregnancy Category C.
For Loteprednol Etabonate had no clinical data on exposed pregnancies are
available. Studies in animals have shown reproductive toxicity. The potential
risk for humans is unknown and it should not be used in pregnancy unless
clearly necessary. It is not known whether topical ophthalmic administration of
corticosteroids could result in sufficient systemic absorption to produce
detectable quantities in human milk. Caution should be exercised when
Loteprednol is administered to a nursing woman.
Precautions & Warnings
If signs and symptoms
fail to improve after two days, the patient should be re-evaluated. If Loteflam
is used for 10 days or longer, intraocular pressure should be monitored. Fungus
invasion must be considered in any persistent corneal ulceration where a
steroid has been used or is in use. If redness or itching becomes aggravated,
the patient should be advised to consult a physician. Patients should also be
advised not to wear contact lenses during their course of therapy.
Therapeutic Class
Ophthalmic Steroid
preparations
Storage Conditions
Store at below 30
degree C in a dry place protected from light. Do not touch tube tip to any
surface. It is desirable that the contents should not be used one month after
first opening of the tube. Protect from freezing.
Frequently Bought Products
Enocam Tablet (10 tablets Strip), Tenoxicam 20 mg
Climycin Capsule ,Clindamycin 300 mg
Montene Tablet, Montelukast Sodium 10 mg
Prebalin CR Tablet (Extended Release), Pregabalin 165 mg
Floxapen Capsule, Flucloxacillin Sodium 250 mg
Methigic Tablet., Methylprednisolone 16 mg
Fulspec IV Injection or Infusion 1 gm Vial, Meropenem Trihydrate 1 gm/vial
Product Queries (0)
Login Or Registerto submit your questions to seller
Other Questions
No none asked to seller yet
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00