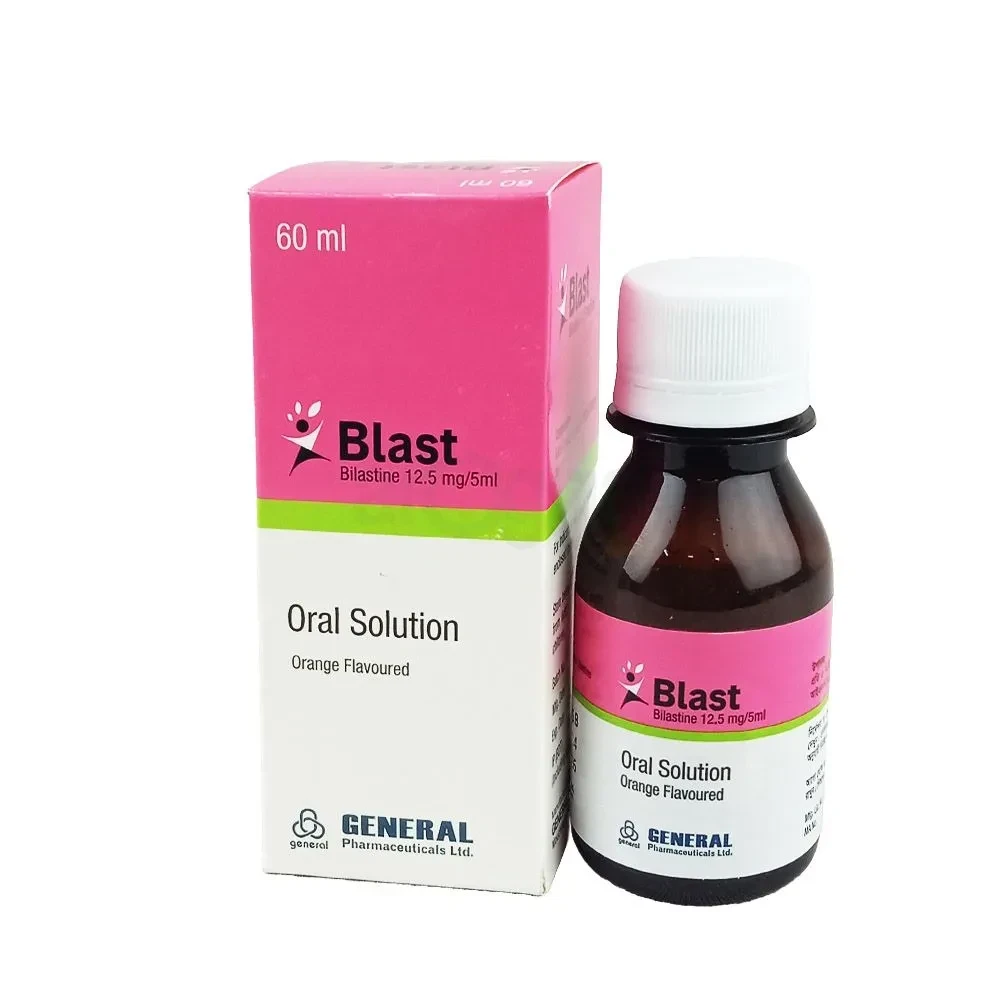

Blast Oral Solution 60 ml bottle, Bilastine 12.5 mg/5 ml
Inhouse product
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00
Reviews & Ratings
Indications
Blast is indicated for
the symptomatic treatment of-
- Allergic rhino conjunctivitis
(seasonal and perennial) and
- Urticaria
Blast is also used to
relieve the symptoms of hay fever (sneezing, itchy, runny, blocked-nose and red
and watery eyes).
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Pharmacology
Bilastine is a potent,
effective, non-sedating, long-acting histamine antagonist with selective & high
affinity to H1 receptor (3 times higher than Cetirizine and 5 times higher than
Fexofenadine). Even at a high concentration, Bilastine does not show affinity
for the 30 other receptors including muscarinic, serotonergic, dopaminergic and
noradrenergic receptors, for the other histamine receptor subtypes (H2, H3 and
H4). It shows excellent safety profile and very favorable pharmacokinetic
characteristics. Bilastine doesn’t undergo any metabolism to be active.
Bilastine is excreted by feces (non -systemic) & urine (systemic)
approximately 66.35% & 28.31% respectively.
Dosage &
Administration
Adults &
adolescents (12 years of age and over): 20 mg tablet once daily for symptomatic relief of allergic
rhinitis, urticaria and allergic rhinoconjunctivitis. The maximum recommended
daily dose is 20 mg Bilastine (1 tablet) and should not be exceeded. If a dose
is missed, the next scheduled dose should be taken. An extra dose should not be
taken. 20 mg Bilastine tablet (1 tablet) once daily should be swallowed with
water on an empty stomach to achieve optimal exposure to Bilastine.
Children between 6 to
11 years: 10 mg mouth
dissolving tablet for the symptomatic relief of allergic rhinitis, allergic
rhinoconjunctivitis and urticaria. The Mouth dissolving tablet is for oral use
only. It should be placed in the mouth. It will disperse rapidly in saliva and
can be easily swallowed. Alternatively, the mouth dissolving tablet can be
dispersed in a tea spoon of water before being swallowed by the children. The
maximum recommended daily dose for children in between 6 to 11 years is 10 mg
Bilastine mouth dissolving tablet (1 tablet) and should not be exceeded. If a
dose is missed, the next scheduled dose should be taken. An extra dose should
not be taken.
Children between 2 to
11 years: 4 ml once daily.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Interaction
Concomitant use of
Blast with ketoconazole, erythromycin, cyclosporine or diltiazem increases the
concentration of Blast. But these changes do not appear to affect the safety
profile of any of the drugs. Intake of alcohol and 20 mg Blast shows same
psychomotor performance similar to that of alcohol and placebo. Concomitant
intake of Blast 20 mg and lorazepam 3 mg for 8 days did not potentiate the
depressant CNS effects of lorazepam.
Contraindications
Bilastine is
contraindicated in patients with hypersensitivity to the active substance or to
any of the excipients of the tablet.
Side Effects
The most commonly
reported side effects in clinical trial are headache, dizziness, somnolence and
fatigue. These adverse events occurred with a comparable frequency in patients
receiving placebo.
Pregnancy &
Lactation
There are no or
limited amount of data from the use of Bilastine in pregnant women. Animal
studies do not indicate direct or indirect harmful effects with respect to
reproductive toxicity, parturition or postnatal development. As a precautionary
measure, it is preferable to avoid the use of Bilastine during pregnancy. The
excretion of Bilastine in milk has not been studied in humans. A decision must
be made taking into account the benefit of breast-feeding for the child and the
benefit of Bilastine therapy for the mother.
Precautions &
Warnings
Treatment with Blast
20 mg does not affect the driving performance. However, patients should be
informed that very rarely some people experience drowsiness, which may affect
their ability to drive or use machines. In clinical trials elderly patients
(≥65 years) showed no difference in effcacy or safety with respect to younger
patients. The maximum plasma concentration of Blast after administration of 20
mg in patients with severe renal impairment is below the safety threshold of
most common adverse e?ects and cardiac or CNS safety. No dosage adjustment is
necessary in patients with renal impairment. Blast is not metabolized in human.
Since renal elimination is the major excretion, biliary excretion is expected
to be only marginally involved in the elimination of Blast. Changes in liver
function are not expected to have a clinically relevant influence.
Use in Special
Populations
Efficacy and safety of
Blast in children under 2 years of age have not been established and there is
little clinical experience in children aged 2 to 5 years, therefore Blast
should not be used in these age groups.
Overdose Effects
In clinical trials,
after administration of Blast at doses 10 to 11 times the therapeutic dose (220
mg as single dose; or 200 mg/day for 7 days) frequency of treatment-emergent
adverse events was two times higher than with placebo. The adverse reactions most
frequently reported were dizziness, headache and nausea. No serious adverse
events and no significant prolongation in the QTc interval were reported.
Therapeutic Class
Non-sedating
antihistamines
Storage Conditions
Keep below 30°C
temperature, protected from light and moisture. Keep out of reach of children.
Chemical Structure
|
Molecular Formula : |
C28H37N3O3 |
|
Chemical Structure : |
|
Common Questions about Blast 12.5 mg/5 ml Oral
Solution
What is Blast 12.5 mg/5 ml Solution?
Blast 12.5 mg/5 ml Solution is H1 receptor
antagnist. It is commonly used to treat Allergic rhino conjunctivitis (seasonal
and perennial) and Urticaria.
Are there any pregnancy warnings of Blast 12.5
mg/5 ml Solution?
Limited information is available. Blast 12.5
mg/5 ml Solution is recommended to be used only after consulting a doctor for
pregnant women.
Are there any breast-feeding warnings of Blast
12.5 mg/5 ml Solution?
It is advised to consult your doctor before
taking Blast 12.5 mg/5 ml Solution if you are breast feeding a baby as relevant
information is not available.
Does Blast 12.5 mg/5 ml Solution safe with
alcohol?
Excessive intake of alcohol should be avoided
during the course of medication as it can increase sleepiness.
Does Blast 12.5 mg/5 ml Solution affect kidney
function?
It is important to consult a doctor to know
the dose adjustment for patients suffering from kidney diseases.
Does Blast 12.5 mg/5 ml Solution affect liver
function?
Relevant information is not available. Please
consult to know if any dose adjustment is needed in patients with liver
impairment.
Quick Tips
- Your doctor has prescribed
Blast 12.5 mg/5 ml Solution to help relieve allergy symptoms such as
itching, swelling, and rashes.
- As compared to other similar
medications, it is much less likely to make you feel sleepy.
- Be cautious while driving or
doing anything that requires concentration as it can cause dizziness and
sleepiness.
- Do not drink alcohol while
taking this medication as it may cause increased sleepiness.
- Stop taking Blast 12.5 mg/5 ml
Solution at least three days before taking an allergy test as it can
affect the test results.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Frequently Bought Products
Cetisoft Capsule, Cetirizine Hydrochloride 10 mg
Loteflam Ophthalmic Gel 3 ml tube, Loteprednol Etabonate 0.5%
Anzitor EZ Tablet, Atorvastatin + Ezetimibe 10 mg+10 mg
Happysol Nasal Drop 10 ml Drop, Sodium Chloride 0.9%
Genacyn Ointment 10 gm tube, Gentamicin Sulfate 0.1%
Product Queries (0)
Login Or Registerto submit your questions to seller
Other Questions
No none asked to seller yet
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00